 
Atomic timeline models must also include an illustration (hand-drawn, picture, or graphic art) and brief summary of Rutherford’s gold foil experiment. Atomic timeline models must include illustrations (hand-drawn, picture, or graphic art) of each scientist’s concept of an atom. Scientist’s main ideas, concept of the atom, or major contribution (paraphrased, can be in bullet form) 3. Atomic timeline models must include the following text/information: a. Atomic timeline models must include the ideas of the following scientists: a. Briefly describe what an orbital is.Ītomic Theory Timeline Assignment Purpose: Create a visual timeline showing the evolution of our understanding of the atom. Modern Model of the Atom: Go to 17.In the mid 1920’s, de Broglie and Schrodinger suggested that electrons have properties of both…ġ8.The current model of the atom depicts electrons to be located in regions called orbitals. what was his “revolutionary idea” about electrons)?Ĭhadwick (and Rutherford): Go to and use the information found there to answer the following questions: 13.What makes up the atomic number?ġ5.What observation led Chadwick (and Rutherford) to conclude there must be something besides just the proton in the nucleus of atoms?ġ6.What is the something-besides-just-the proton called? Use the information found there to answer the following questions: 10.Describe Rutherford’s Gold Foil Experimentġ1.How much smaller was the nucleus, than the atom itself, according to Rutherford? Sketch a picture of Rutherford’s model of the atomġ2.How did Bohr modify the plum pudding model of the atom (i.e. 
Rutherford and Bohr Break the “Plum Pudding” Model: Go to. What was the model of the atom he proposed in 1904? Sketch a picture of this model What was the evidence for “bodies much smaller than atoms”?ĩ. What was the name of the experiment in which Thomson discovered the electron?Ĩ. Thomson: Go to and use the information there to answer the following questions: 6. What are the four components of Dalton’s Atomic Theory? When did Dalton form his Atomic Theory (often known as the Billiard Ball or Solid Sphere Model)?ĥ. John Dalton’s Atomic Theory: Go to and use the information there to answer the following questions: 4. How were the ideas of these two men received by Aristotle, and what was the result on the progress of atomic theory for the next 2,000 years? 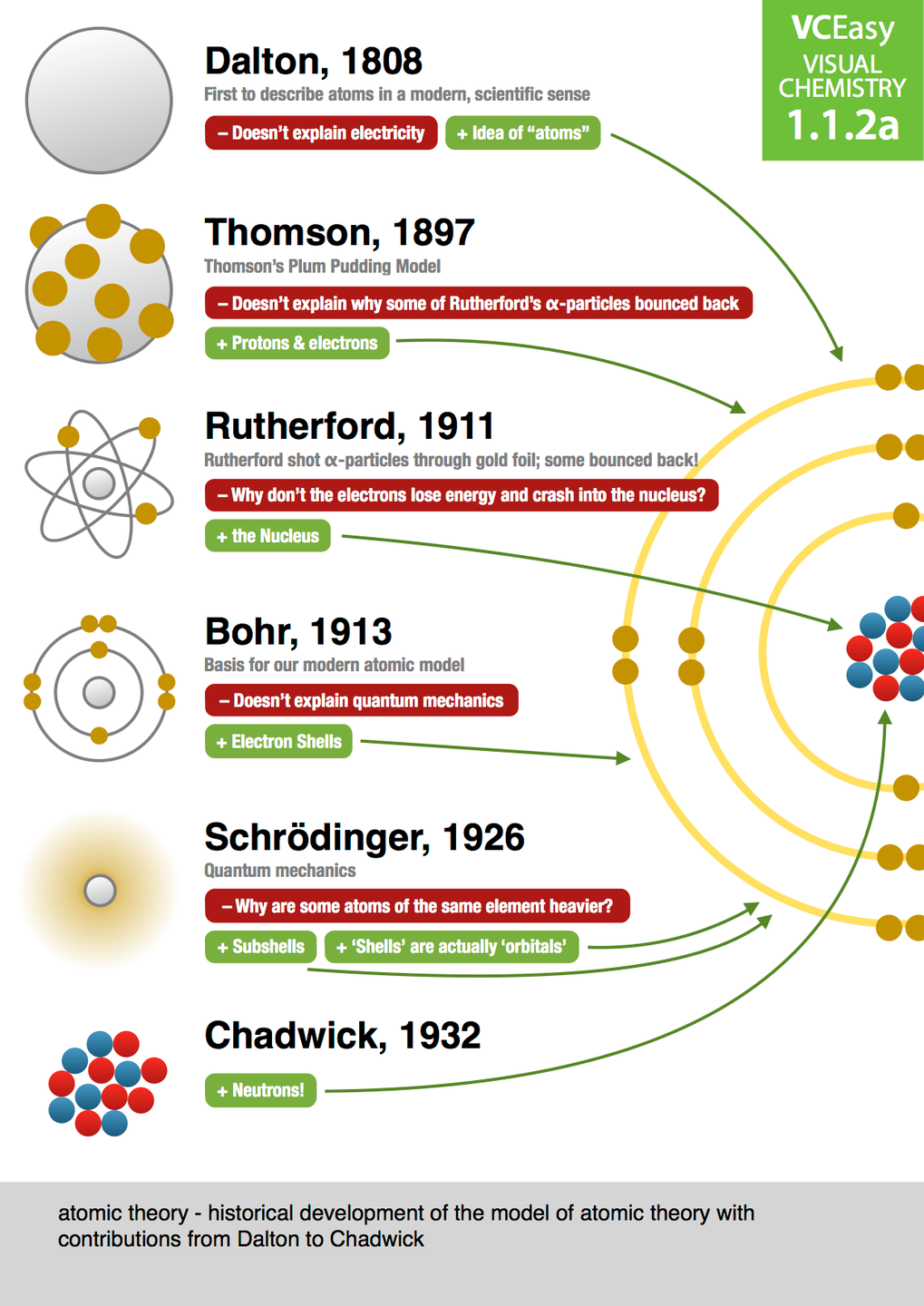
How did they use atoms to explain different physical properties?ģ. What was the “basic idea” about matter that Leucippus and Democritus proposed?Ģ. Early Ideas About Atoms: Go to and read the section on “Early Greek Ideas” in order to answer the following questions: 1. Webquest: Atomic Theories and Models Answer the following questions regarding the progression of the model of the atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
